New and used medical equipment listings are published daily at MedGlobalGroup.com!🌎https://loom.ly/NFdarqk #medicalequipment #medicaldevices #biomedical #medicaldevicesales #ventilator

FlexiMagPlus | MedGlobal Group
Intelligent synchrony in mechanical ventilation. High Performance Ventilation for your ICU
https://loom.ly/NFdarqkNew and used medical equipment listings are published daily at MedGlobalGroup.com!🌎https://loom.ly/NFdarqk #medicalequipment #medicaldevices #biomedical #medicaldevicesales

FlexiMagPlus | MedGlobal Group
Intelligent synchrony in mechanical ventilation. High Performance Ventilation for your ICU
https://loom.ly/NFdarqkNew and used medical equipment listings are published daily at MedGlobalGroup.com!🌎https://loom.ly/NFdarqk #medicalequipment #medicaldevices #biomedical #medicaldevicesales

FlexiMagPlus | MedGlobal Group
Intelligent synchrony in mechanical ventilation. High Performance Ventilation for your ICU
https://loom.ly/NFdarqkhttps://www.medtechdive.co...

FDA breakthrough device designations fall in Q3 but record year still within reach
MedTech industry news
https://www.medtechdive.com/news/fda-breakthrough-device-designations-fall-q3/635301/?utm_source=Sailthru&utm_medium=email&utm_campaign=Issue:%202022-10-31%20MedTech%20Dive%20%5Bissue:45645%5D&utm_term=MedTech%20DiveBonjour,
- FDA publishes GDUFA III user fees, increases ANDA, DMF fees.
- Merck’s cardiovascular disease drug succeeds in late-stage trial, validates US$ 11.5 bn Acceleron buyout.
- FDA grants fast-track status to Lilly’s diabetes drug Mounjaro to treat obesity.
- Bayer’s Vividion signs potential US$ 930 million oncology deal with Tavros.
- GSK’s Boostrix vaccine gets add-on approval to prevent whooping cough in infants.
- Sanofi, Regeneron flaunt Dupixent’s benefit in eosinophilic esophagitis trial in kids.
- Two-year data on Roche’s Evrysdi shows benefit in spinal muscular atrophy.
- US ADHD drug crisis deepens as Novartis’ Sandoz also reports shortage of Adderall.
- EDQM finds one major deficiency at Biocon’s API facility in Bengaluru.
#Phispers This Week - PharmaCompass.com's Weekly News Recap
#Healthcare #pharmaceuticals #pharmanews #pharmaceuticalindustry
https://lnkd.in/dE5X4qag

FDA steps up user fee under GDUFA III; Merck’s heart drug succeeds in late-stage study | Radio Compass Phisper
On September 30, the US President Joe Biden had signed into law the Food and Drug Administration Reauthorization Act (FDARA), which includes the reauthorization of the Generic Drug User Fee Amendments (GDUFA) through September 2027. In this week’s Phispers, we bring you the new GDUFA III fe...
https://lnkd.in/dE5X4qaghttps://www.medglobalgroup...
Bata De Aislamiento De Nivel 2 Sms (No Estéril) – Aprobado Por Fda/Ce | MedGlobal Group
https://www.medglobalgroup.com/es/product/level-2-isolation-gown-sms-non-sterile-fdace-approved

New Treatment Options in Oncology: FDA and EMA Drug Approvals in Q3 2022
Aptitude Health’s quarterly recap of FDA- and EMA-approved oncology agents: Q3 2022 focus on novel agents and expanded indications
https://lnkd.in/e57jvaAuby John R. Fischer, Senior Reporter |
-
Amyloid PET scans can accurately diagnose or rule out Alzheimer’s disease in veterans with memory loss and may be useful for managing their long-term clinical care.
That’s what researchers in the Memory Disorders Clinic at the VA Boston Healthcare System found in two studies on the scans, which are minimally invasive diagnostic procedures for measuring amyloid, a naturally occurring protein in and common biomarker of the disease.
The clinic is one of the few VA programs where this type of imaging is conducted, with others and private sector facilities not typically performing it, because of insurers and Medicare not reimbursing for it, despite being FDA-approved.
Read more at https://www.dotmed.com/new...
Amyloid PET scans can rule out Alzheimer's, inform long-term care planning
Two studies assessed 565 veterans
https://www.dotmed.com/news/story/58928By Kaitlyn Wilkie
-
Improving image quality and dose efficiency have always been top priorities in the development of new imaging systems to improve diagnostics and patient care. Photon-counting detectors (PCDs) are the newest major advancement for CT systems. These detectors have already been used for several years in nuclear imaging.
In September 2021, Siemens Healthineers received FDA approval for the NAEOTOM Alpha, the first photon-counting CT system cleared for the U.S. market. Neurologica received FDA approval for its photon-counting CT system, the OmniTom Elite with PCD, in March 2022. What makes these systems unique among other CT systems on the market is the PCD, which allows for spectral imaging. Conventional CT systems use third-generation digital radiography (DR) detectors. The PCD’s advantages are its ability to provide increased spatial resolution, decrease electronic noise, and reduce radiation exposure compared to DR detectors.
Read more at https://www.dotmed.com/new...

Photon-counting detectors advance CT technology
Market insight from the expert analysts at symplr
https://www.dotmed.com/news/story/58825https://www.youtube.com/wa...
Biomeds are drastically hindered when we are not provided the bare minimum that anyone attempting to fix any device would require. We need access to parts, access to service manuals, and the possibility to be provided training. If OEM's are so concerned we cannot do the level of work to their standard then provide us the training so we meet your standards.
The FDA report mentioned that shows third party biomed services are a vital component to the medial device service landscape.
Bonjour,
- FDA approves world’s costliest therapy, priced at US$ 3 million, from Bluebird bio.
- Mallinckrodt receives FDA approval to treat life-threatening liver condition.
- Novartis to move Supreme Court to secure validity of Gilenya patent.
- FDA issues Form 483 with 12 observations to Catalent’s Indiana site.
- BMS, AbbVie to lay off up to 360 employees in California.
- BMS’ Opdivo shows promise as adjuvant treatment for melanoma.
- EMA recommends AstraZeneca-Sanofi’s RSV drug for infants.
#Phispers in a minute - PharmaCompass.c
https://www.prnewswire.com...
#wearables #remotepatientmonitoring #digitalhealth #telehealth #FDA #medicaldevices #military #veterans #veteransaffairs #VA #health #wellness #safety #accuracymatters #ethicsmatter
…see more
Dr. Robert A.
Dr. Robert A.
• 2nd
3d • Edited • 3 days ago
Follow
✅ "We were very impressed by Inviza®Health — Powering Telehealth Connectivity™'s footstep #charging , SmartHealth shoe #insole #wearabletechnology
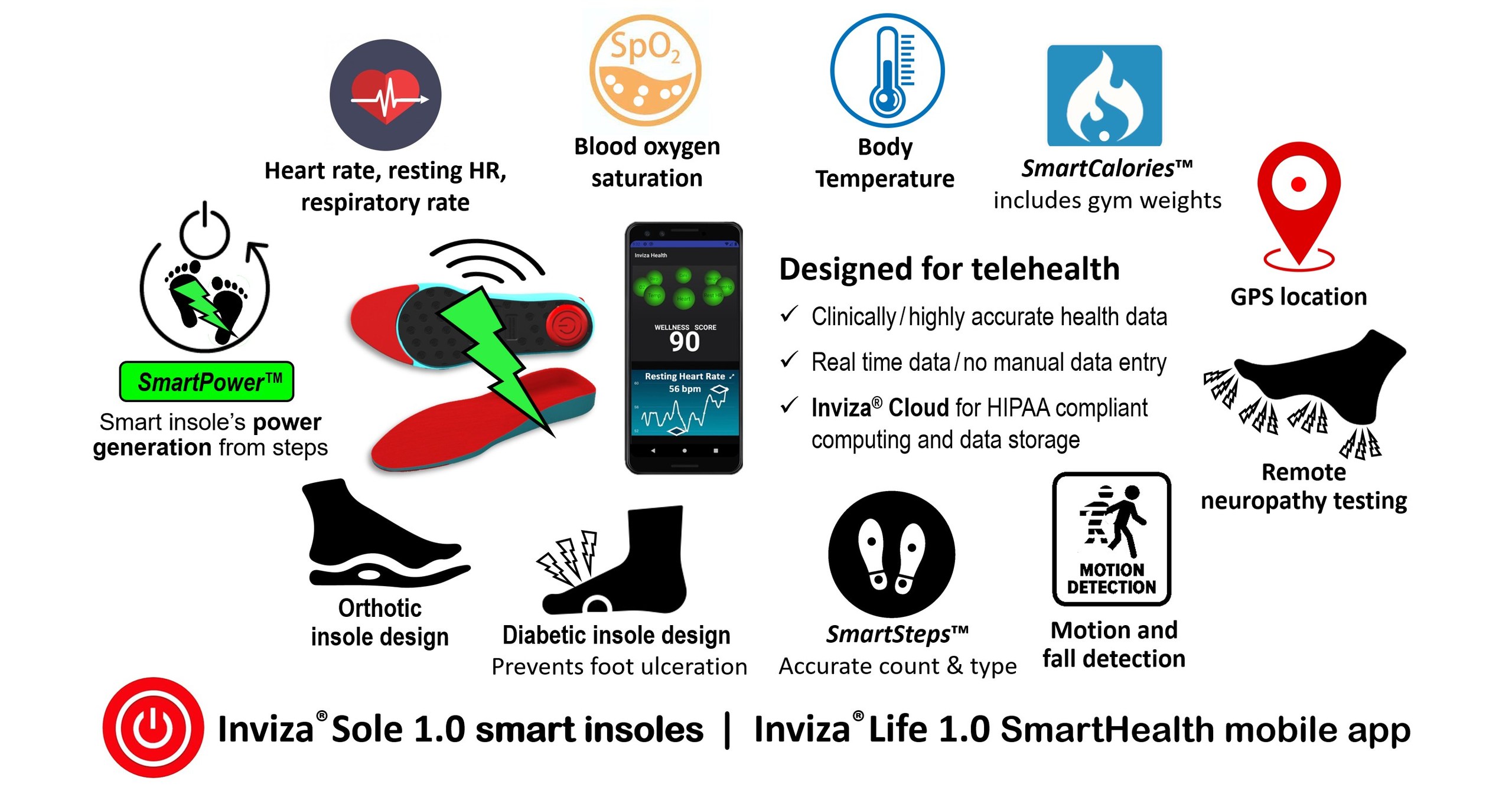
Inviza® Showcases World's 1st Energy Harvesting Powered Digital Health Shoe Insoles to U.S. Air Force
/PRNewswire/ -- Inviza® Health was invited to the inaugural Innovare Advancement Center MedTech Workshop at their Internet of Things (IoT) Living Laboratory on...
https://www.prnewswire.com/news-releases/inviza-showcases-worlds-1st-energy-harvesting-powered-digital-health-shoe-insoles-to-us-air-force-301630018.htmlA couple of weeks ago, we got FDA Clearance for the world’s
first, high-power LED device for permanent hair reduction, Epi NIR!!
We are now looking for successful INDEPENDENT US SALES REPRESENTATIVE to help us sell and market Epi NIR in the US.
Contact us at infogloballaunch.com
or Tom Gordon at tgordonlasersyahoo.com
or Bill Hough at billgloballaunch.com
#aesthetics #medical #medicalaesthetics #aestheticsdevice #device #medicaldevice #esthetics #ethetician #plasticsurgeon #plasticsurgery #medspa #medicalspa #medspas #hair #ha
- FDA approves world’s costliest therapy, priced at US$ 3 million, from Bluebird bio.
- Mallinckrodt receives FDA approval to treat life-threatening liver condition.
- Novartis to move Supreme Court to secure validity of Gilenya patent.
- FDA issues Form 483 with 12 observations to Catalent’s Indiana site.
- BMS, AbbVie to lay off up to 360 employees in California.
- BMS’ Opdivo shows promise as adjuvant treatment for melanoma.
- EMA recommends AstraZeneca-Sanofi’s RSV drug for infants.
#Phispers This Week - PharmaCompass.com's Weekly News Recap
#Healthcare #pharmaceuti

Bluebird ousts its own gene therapy approved last month as world’s costliest; Catalent hit by FDA’s Form 483 | Radio Compass Phisper
Exactly a month after Bluebird bio’s gene therapy Zynteglo became the most expensive treatment at US$ 2.8 million, a second therapy from the drugmaker has clinched that title. The drug – Skysona or eli-cel – approved by the US Food and Drug Administration (FDA) to treat a rare n...
https://lnkd.in/dkGiTYgm

